- Your cart is empty
- Continue Shopping
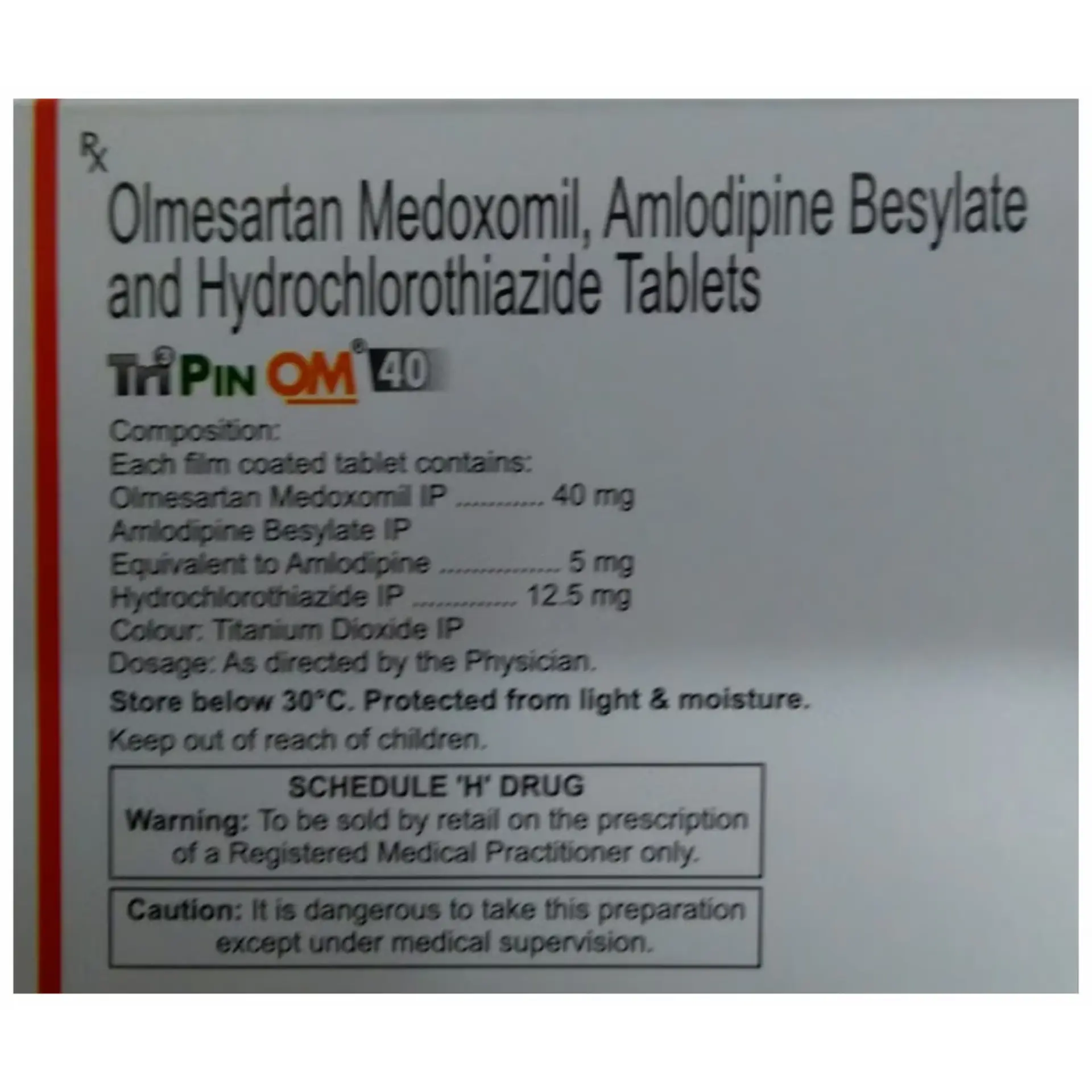
Tripin OM 40 Tablet is formulated as a combination therapy to manage hypertension, particularly in cases where monotherapy does not yield satisfactory results. By effectively lowering blood pressure, this medication plays a significant role in decreasing the risk of serious cardiovascular incidents such as heart attacks and strokes. The administration of Tripin OM 40 Tablet can be done either with or without food, and the dosage will be determined based on the individual’s medical condition and response to treatment. It is important to take this medication consistently at the same time each day to maintain stable blood pressure levels. Patients are advised to continue their course of treatment until directed otherwise by their healthcare provider. This medication includes a diuretic component, which may lead to increased urination; therefore, patients should avoid taking it close to bedtime to minimize nighttime disturbances. For optimal results, regular intake is crucial, even if one feels well since hypertension often presents without symptoms. Discontinuing treatment could elevate the likelihood of heart-related complications. Staying hydrated is recommended to alleviate potential side effects such as muscle weakness, dry mouth, and excessive thirst. Regular monitoring of blood pressure, kidney function, electrolyte levels, and blood sugar is essential to ensure the medication is effective and safe.






Reviews
There are no reviews yet.